Abstract
Objective: To evaluate the benefits and the medium-term side effects of methylprednisolone in very preterm infants at risk of chronic lung disease.
Study design: Forty-five consecutive preterm infants (<30 weeks' gestation) at risk of chronic lung disease were treated at a mean postnatal age of 16 days with a tapering course of methylprednisolone. The outcome of treatment was assessed by comparison with 45 consecutive historical cases of infants treated with dexamethasone; the infants did not differ in baseline characteristics.
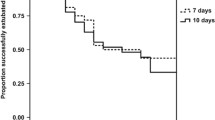
Results: There were no differences between groups in the rate of survivors without chronic lung disease. Infants treated with methylprednisolone had a higher rate of body weight gain during the treatment period (median 120 g, range 0 to 190, vs. 70 g, range –110 to 210, P=0.01) and between birth and the age of 40 weeks (median 1660 g, range 1170–2520, vs. 1580 g, range 1040 to 2120, P=0.02). The incidence of both glucose intolerance requiring insulin (0% vs. 18%, P=0.006) and cystic periventricular leukomalacia (2% vs. 18%, P=0.03) was lower among methylprednisolone-treated infants.
Conclusion: Our observations confirm methylprednisolone to be as effective as dexamethasone and to have fewer side effects. A randomized control trial is needed to further study the efficacy and safety of methylprednisolone in very premature infants at risk of chronic lung disease.
Article PDF
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Final revision received: 19 May 2000
Electronic Publication
Rights and permissions
About this article
Cite this article
André, P., Thébaud, B., Odièvre, M. et al. Methylprednisolone, an alternative to dexamethasone in very premature infants at risk of chronic lung disease. Intensive Care Med 26, 1496–1500 (2000). https://doi.org/10.1007/s001340000588
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s001340000588