Abstract
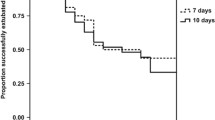
Short term inhaled dexamethasone therapy was evaluated in a double blind placebo controlled trial in 36 ventilator dependent preterm neonates (BW<1500 gm, postnatal age>7 days) who were at risk for bronchopulmonary dysplasia. Pulmonary and systemic effects were compared at early (day 3), late (7–10 days) and post (14 days after initiation) phases of therapy. Airflow mechanics improved as demonstrated by a net 101% improvement in pulmonary resistance (a decrease from 139 to 101 cm H2O/L/s in the dexamethasone treated infants as compared to an increase from 153 to 267 cm H2O/L/s in the placebo treated infants during the early phase of therapy); this was associated with a 45% increase in inspiratory airflow (1.29±0.43 to 1.87±0.978 L/min; p<0.01), and 37% increase in expiratory airflow. These changes resulted in a significant reduction in the work of breathing such that the mean tidal driving pressure significantly decreased from 13.6 cmH2O to 9.4 cm H2O with inhaled steroid administration. Though the brief duration of therapy did not result in cessation of ventilatory support, the level of support was significantly reduced (decreased values of oxygen supplementation, mean airway pressure and oxygenation index and increased ventilatory efficiency index). The inhaled dexamethasone therapy was also associated with systemic absorption of the drug as evidenced by transient but apparently reversible reduction in serum cortisol levels. No systemic side effects of hypertension, hyperglycemia or nosocomial sepsis were observed. These data demonstrate beneficial effects of short term inhaled dexamethasone on the resistive airflow properties of preterm infants at risk for BPD and may provide adjunctive means to facilitate weaning in the ventilator dependent neonates.
Article PDF
Similar content being viewed by others
References
Avery GB, Fletcher AB, Kaplan M, Brudno DS. Controlled trial of dexamethasone in respirator-dependent infants with bronchopulmonary dysplasia.Pediatrics 1985; 75: 106–111.
Cummings JJ, D'Eugenio DB, Gross SJ. A controlled trial of dexamethasone in preterm infants as high risk for broncho-pulmonary dysplasia.New Engl J Med 1989; 320: 1505–1510.
Collaborative Dexamethasone Trial Group. Dexamethasone therapy in neonatal chronic lung disease: An international placebo-controlled trial.Pediatrics 1991; 88: 421–427.
Yeh TF, Torre JA, Rostogi A, Anyebuno MA, Pildes RS. Early postnatal dexamethasone therapy in premature infants with severe respiratory distress syndrome: a double blind controlled study.J Pediatr 1990; 117: 273–282.
Harkavy KL, Scanlon JW, Chowdhry PK, Grylack LJ. Dexamethasone therapy for chronic lung disease in ventilator and oxygen-dependent infants: A controlled trial.J Pediatr 1989; 115: 979–983.
Ohlsson A, Calvert S, Hosking M, Shennan A. Randomized controlled trial of dexamethasone treatment in very low birth weight infants with ventilator dependent chronic lung disease.Pediatr Res 1989; 25: 225A.
Mammel MC, Johnson DE, Green TP, Thompson TR. Controlled trial of dexamethasone therapy in infants with broncho-pulmonary dysplasia: Acute effects and 1-year follow-up.Dev Pharmacol Ther 1987; 10: 1–11.
LaForce WR, Brudno DS. Controlled trial of beclomethasone dipropionate by nebulization in oxygen and ventilator-dependent infants.J Pediatr 1993; 122: 285–258.
Bhutani VK, Abbasi S. Relative likelihood of bronchopulmonary dysplasia based on pulmonary mechanics measured in preterm neonates during the first week of life.J Peds 1992; 120: 605–613.
Bhutani VK, Abbasi S, Sivieri EM, Fox WW, Shaffer TM. Evaluation of neonatal pulmonary mechanics and energetics: A two factor least mean square analysis.Pediatr Pulmonol 1988; 4: 150–158.
Pappagallo M, Blondheim O, Bhutani VK, Abbasi S. Effect of inhaled dexamethasone in ventilator dependent preterm infants.Pediatr Res 1990; 27: 219A.
Noble-Jamieson CM, Regev R, Silverman M. Dexamethasone in neonatal chronic lung disease: Pulmonary effects and intra-cranial complications.Eur J Pediatr 1989; 148: 365–367.
Williams TJ, Yarwood H. Effect of glucocorticosteroids on microvascular permeability.Am Rev Respir Dis 1990; 141: S39–43.
Schleimer RP, Freeland HS, Peters SP, Brown KE, Derse CP. An assessment of the effects of glucocorticoids on degranulation, chemotaxis, binding to vascular endothelium and formation of leukotriene B4 by purified human neutrophils.J Pharmacol Exp Ther 1989; 250: 598–605.
Groneck P, Reuss D, Gotze-Speer B, Speer CP. Effects of dexamethasone on chemotactic activity and inflammatory mediators in tracheobronchial aspirates of preterm infants at risk for chronic lung disease.J Pediatr 1993; 122: 938–44.
Finer NN, Etches PC. Fiberoptic bronchoscopy in the neonate.Pediatr Pulmonol 1989; 7: 116–20.
Abbasi S, Silva W, Gerdes JS, Bhutani VK. Role of dexamethasone therapy for severe tracheobronchial mucosal lesions in mechanically ventilated preterm infants.Ped Res 1993; 33: 198A.
Alkalay AL, Pomerance JJ, Puri AR,et al. Hypothalamic-pituitary-adrenal axis function in very low birth weight infants treated with dexamethasone.Pediatrics 1990; 86: 204–210.
Wilson D, Baldwin R. A randomized placebo controlled trial of effects of dexamethasone on hypothalamic pituitary adrenal axis in preterm infants.J Pediatr 1988; 113: 764–768.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pappagallo, M., Abbasi, S. & Bhutan, V.K. Respiratory and systemic effects of inhaled dexamethasone on ventilator dependant preterm infants at risk for bronchopulmonary dysplasia. Indian J Pediatr 65, 273–282 (1998). https://doi.org/10.1007/BF02752304
Issue Date:
DOI: https://doi.org/10.1007/BF02752304