Article Text
Abstract
Objective To assess the effect of volume-targeted ventilation (VTV) compared with pressure-limited ventilation (PLV) in preterm infants.
Method We searched the Cochrane Library (Issue 3, 2013), PubMed (1966 to 5 March 2013), China National Knowledge Infrastructure (CNKI) and periodical databases (1979 to 5 March 2013). We selected randomised controlled trials (RCTs) and quasi-RCTs of VTV versus PLV as active interventions in preterm infants. We performed meta-analyses using the Cochrane statistical package RevMan 5.0.
Results Eighteen trials met our inclusion criteria. There was no evidence that VTV modes reduced the incidence of death (relative risk (RR) 0.73, 95% CI 0.51 to 1.05). The use of VTV modes resulted in a reduction in the incidence of bronchopulmonary dysplasia (BPD) (RR 0.61, 95% CI 0.46 to 0.82) and duration of mechanical ventilation (mean difference (MD) −2.0 days, 95% CI −3.14 to −0.86). VTV modes also resulted in reductions in intraventricular haemorrhage (IVH) (RR 0.65, 95% CI 0.42 to 0.99), grade 3/4 IVH (RR 0.55, 95% CI 0.39 to 0.79), periventricular leukomalacia (PVL) (RR 0.33, 95% CI 0.15 to 0.72), pneumothorax (RR 0.52, 95% CI 0.29 to 0.93), failure of primary mode of ventilation (RR 0.64, 95% CI 0.43 to 0.94), hypocarbia (RR 0.56, 95% CI 0.33 to 0.96), mean airway pressure (MD −0.54 cmH2O, 95% CI −1.05 to −0.02) and days of supplemental oxygen administration (MD −1.68 days, 95% CI −2.47 to −0.88).
Conclusions Preterm infants ventilated using VTV modes had reduced duration of mechanical ventilation, incidence of BPD, failure of primary mode of ventilation, hypocarbia, grade 3/4 IVH, pneumothorax and PVL compared with preterm infants ventilated using PLV modes. There was no evidence that infants ventilated with VTV modes had reduced death compared to infants ventilated using PLV modes.
- Intensive Care
- Neonatology
Statistics from Altmetric.com
Introduction
Respiratory support in the neonatal intensive care unit continues to evolve rapidly. However, preterm lungs are particularly susceptible to ventilator-induced lung injury (VILI).1–3 VILI is a major contributor to chronic lung disease (CLD) such as bronchopulmonary dysplasia (BPD). BPD is a persisting complication among surviving preterm infants and its incidence has not decreased in the last decade.4 BPD causes significant complications in the newborn and is associated with mortality, cardiopulmonary dysfunction, re-hospitalisation, growth failure and poor neurodevelopmental outcome following hospital discharge. There are many possible factors involved in the pathogenesis of BPD, including oxygen toxicity, volutrauma and infection.5–7
Traditional time-cycled, pressure-limited modes of mechanical ventilation (pressure-limited ventilation (PLV)) ventilate effectively due to the decelerating gas-flow pattern and the presumed benefit of directly controlling peak inspiratory pressure (PIP).8 The major disadvantage of PLV lies in the variable tidal volume that results from changes in lung compliance. The consequences of improvements in compliance are inadvertent hyperventilation and lung damage from excessively large tidal volumes. Avoiding excessive tidal volume is essential for limiting lung injury.9–11
Volume-targeted ventilation (VTV) strategies are modifications of PLV that employ different microprocessor-driven algorithms to adjust inspiratory pressure, flow or time to achieve a target tidal volume. VTV is increasingly used in extremely low birthweight infants because of the potential for reducing volutrauma and avoiding hypocapnia. Clinical trials indicated a decrease of PIP and mean airway pressure (MAP) when comparing VTV with conventional PLV modes in very low birthweight infants recovering from acute respiratory failure, stable ventilated premature newborns and premature newborn infants with respiratory distress syndrome (RDS).12–14
A recent Cochrane review comparing VTV versus PLV in the neonate concluded infants ventilated using VTV modes had reduced death and CLD compared with infants ventilated using PLV modes.15 ,16
The aim of our review is to further assess the effect of VTV compared with PLV in preterm infants and update the Cochrane review with evidence from an additional six studies.
Methods
Inclusion criteria
We included randomised controlled trials (RCTs) and quasi-RCTs comparing VTV, volume guarantee ventilation (VG), volume assured pressure support (VAPS), minimal volume guarantee and pressure-regulated volume control (PRVC) modes of ventilation with PLV. We considered for inclusion, all infants <37 weeks’ gestation and birth weight ≤2500 g receiving artificial ventilation. Primary outcome measures were incidence of death, BPD (BPD was defined as oxygen treatment or respiratory support at 36 weeks’ corrected gestational age) and length of mechanical ventilation. Secondary outcome measures were neurodevelopmental outcomes, pneumothorax, intraventricular haemorrhage (IVH), periventricular leukomalacia (PVL), failure of primary mode of ventilation, oxygen therapy duration, duration of hospitalisation, any episode of hypocarbia (pCO2 <35 mm Hg/4.7 kPa), any episode of hypercarbia (pCO2 >60 mm Hg/8 kPa), overall incidence of air leak, patent ductus arteriosus, neonatal sepsis, second dose of surfactant, pulmonary haemorrhage, retinopathy of prematurity, necrotising enterocolitis and ventilatory parameters.
Search methods for identification of studies
We searched the Cochrane Library (Issue 3, 2013), PubMed (1966 to 5 March 2013), China National Knowledge Infrastructure (CNKI) and periodical databases (1979 to 5 March 2013). We used the keywords: volume-controlled ventilation OR VTV OR volume guarantees ventilation OR PRVC ventilation OR targeted tidal volume OR VAPS OR minimal volume guarantee AND preterm infants OR low birthweight infants.
Selection of studies
Two review authors (Peng WanSheng and Zhu HongWei) independently assessed the titles and abstracts for the selection of all eligible studies identified by the searches. We obtained the full articles when they appeared to meet the inclusion criteria or there were insufficient data in the title and abstract to make a clear decision for their inclusion. We excluded articles that did not meet the inclusion criteria. We noted the reasons for their exclusion. We resolved any disagreements between the two review authors about study inclusion by discussion.
Data extraction and management
Both authors independently extracted data and entered the data into RevMan 5. When data were missing or unclear, we contacted the trial authors for clarification. For dichotomous outcomes, the number of participants experiencing the event and the number assessed in each group were recorded. For continuous outcomes, the arithmetic means, SDs and number assessed in each group were extracted.
Assessment of risk of bias in included studies
Both authors independently assessed the methodological quality of each trial using the Cochrane Collaboration's recommendations.17 Descriptive data were collected on whether participants, care providers or outcome assessors were blinded; sequence generation was adequate; allocation was concealed; incomplete outcome data were clearly addressed; there was selective reporting; there was other reporting. Assessment results are summarised in figure 1.
Quality assessment of the 18 included trials. +, Yes; − No; ?, Unclear.
Statistical analyses
We synthesised dichotomous data using relative risk (RR), number needed to treat (NNT) and 95% CIs as effect measures. We used the mean difference (MD) and 95% CI as the metrics of effect size for continuous outcomes. We assessed heterogeneity in results between studies using the Cochrane Q test (p<0.1 considered significant) and the I2 statistic.
Results
Description of studies
The initial electronic searches retrieved a total of 350 citations. After reviewing the titles and abstracts, we identified 59 papers as being potentially relevant. After reading the full text articles, 18 trials enrolling 954 children met our inclusion criteria.14 ,18–35 The process of trial selection is shown in figure 2.
Flow chart of article screening and selection process. On the basis of the search strategy, 350 articles were identified by the initial electronic search of the medical literature databases and 59 required further assessment. Finally, 19 articles describing 18 trials were included in this review. HFV, high frequency ventilation; RCT, randomised controlled trials; SIMV, synchronised intermittent mandatory ventilation; VG, volume guarantee ventilation; VTV, volume-targeted ventilation.
Trials included
Eighteen studies reporting RCTs met our inclusion criteria (table 1). Of these, 14 were parallel studies, resulting in 15 publications. Four trials were within-patient crossover studies.14 ,22 ,23 ,30 In the crossover trials, duration of intervention period ranged from 60 min up to 4 h. The characteristics of infants included varied between the trials.
Characteristics of the 18 randomised controlled trials included in the meta-analysis
Trials identified, but excluded
The published data of the Salvia and TARDIS trials were not included.36 ,37
Interventions
A range of ventilators delivering VTV were used for the experimental groups, including the VIP Bird, Siemens Servo 300, Bear Cub, Draeger Babylog 8000plus, SLE5000, Sensor Medics 3100A and Sechrist Infant ventilator. Ventilation modes are described in table 1. Ventilation settings were not always well described in each trial.
Risk of bias in included studies
Risk of bias in included studies is summarised in figure 1.
Randomisation pattern
Eleven trials used a block randomisation and random number chart to generate the random sequence.18–21 25–28 ,31 ,33 ,34 The method of generation for random sequence was unclear in seven trials.14 ,29 ,22–24 ,30 ,35
Blinding
Sixteen studies used sealed and opaque envelopes for allocation concealment.18–25 27–34 Assessors of neurodevelopmental outcomes and radiologists were blinded to group assignment in three trials.19 ,32 ,33
Completeness of study outcome data
The number of participants with missing data was small or nil in all trials. Thus, incomplete outcome data may not be a source of bias in this review. There was no evidence of selective reporting of outcomes in 16 trials.18–25 27–34
No other potential sources of bias were observed.18–21 25–28 ,31 ,33 ,34
Results of meta-analysis
Primary outcomes
Incidence of death
Data reporting the incidence of death in VTV and PLV (control) groups (n=767) are described in 11 trials.19–21 24–27 ,29 ,30 ,31 ,32 The meta-analysis demonstrated no significant difference in the incidence of death between VTV and PLV groups (RR 0.73 (95% CI 0.51 to 1.05), p=0.09; figure 3). There was no evidence of significant heterogeneity between trials (p=0.80, I2=0).
Volume-targeted ventilation (VTV) versus pressure-limited ventilation (PLV): incidence of death in hospital. Eleven trials described the incidence of death in the VTV and PLV groups (n=767). The risk ratio was 0.73 (95% CI 0.51 to 1.05, p=0.09). There was no evidence of significant heterogeneity between trials (p=0.80, I2=0). This figure was created by RevMan V5.0.0. M-H, Mantel-Haenszel test.
Incidence of BPD
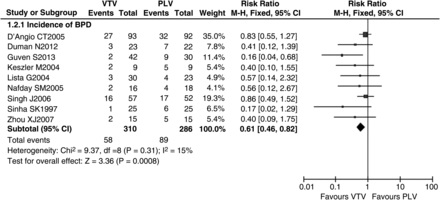
Data reporting the incidence of BPD at 36 weeks’ corrected gestational age in VTV and PLV groups are described in nine trials.19–21 ,24 ,25 ,27 ,31 ,33 ,35 The meta-analysis demonstrated a significantly lower incidence of BPD in preterm infants treated with VTV compared to those treated with PLV (RR 0.61 (95% CI 0.46 to 0.82), p=0.0008; figure 4). NNT=8 (95% CI 5 to 20). There was evidence of low heterogeneity between trials (p=0.31, I2=15%).
Volume-targeted ventilation (VTV) versus pressure-limited ventilation (PLV): incidence of bronchopulmonary dysplasia (BPD). Nine trials described incidence of BPD in VTV and PLV groups (n=596). The meta-analysis showed that incidence of BPD produced significantly different effects in VTV and PLV groups. The risk ratio was 0.61 (95% CI 0.46 to 0.82, p=0.0008). There was no evidence of significant heterogeneity between trials (p=0.31, I2=15%). This figure was created by RevMan V.5.0.0. M-H, Mantel-Haenszel test.
Length of mechanical ventilation
Data reporting the length of mechanical ventilation in VTV and PLV groups are described in nine trials.19 ,21 ,24–26 ,29 ,31 ,33 ,35 The meta-analysis revealed a mathematical MD of −2.0 (−3.14 to −0.86) days reduced duration of ventilation using VTV (figure 5). Two trials reported median values and showed no significant differences between the VTV and PLV groups. There was evidence of significant heterogeneity between studies (p=0.07, I2=45%). We performed subgroup analyses to explore the possible causes of heterogeneity in effect sizes across studies. Subgroup analyses were performed according to ventilation mode and ventilators, birth weight, gestational ages and methodology. There was evidence of statistically significant heterogeneity in the subgroups.
Volume-targeted ventilation (VTV) versus pressure-limited ventilation (PLV): duration of mechanical ventilation. Nine trials described duration of mechanical ventilation in VTV and PLV groups (n=604). The meta-analysis showed that duration of mechanical ventilation produced significantly different effects in VTV and PLV groups. The mean difference was −2.0 days (95% CI −3.14 to −0.86, p=0.0006). There was no evidence of significant heterogeneity between trials (p=0.07, I2=45%). This figure was created by RevMan V5.0.0. IV, inverse variance test.
Secondary outcomes
Incidence of IVH, grade 3/4 IVH and PVL
Data reporting the incidence of IVH in VTV and PLV groups (n=759) are described in 11 trials.19–21 24–27 29–31 ,33 The meta-analysis demonstrated a significantly lower incidence of IVH in preterm infants treated with VTV compared to those treated with PLV (RR 0.65 (95% CI 0.42 to 0.99), p=0.04). There was evidence of moderate heterogeneity between studies (p=0.02, I2=53%). Subgroup analysis was performed according to ventilation mode and ventilators, birth weight, gestational ages and methodology. There was no evidence of heterogeneity in the subgroup ventilated with similar modes (I2=0), but there was evidence of significant heterogeneity in the subgroup ventilated with different modes (I2=54%).
Data reporting the incidence of grade 3/4 IVH in VTV and PLV groups are described in 10 trials.19–21 24–27 29–31 The meta-analysis demonstrated a significantly lower incidence of grade 3/4 IVH in preterm infants treated with VTV compared to those treated with PLV (RR 0.55 (95% CI 0.39 to 0.79), p=0.001). NNT was 11 (95% CI 7 to 25). There was no evidence of significant heterogeneity between studies (p=0.32, I2=14%).
Data reporting the incidence of PVL in VTV and PLV groups are described in seven trials.19–21 ,24 ,25 ,31 ,33 The meta-analysis demonstrated a significantly lower incidence of PVL in preterm infants treated with VTV compared to those treated with PLV (RR 0.33 (95% CI 0.15 to 0.72), p=0.006). NNT was 16 (95% CI 10 to 50). There was no evidence of significant heterogeneity between studies (p=0.92, I2=0).
Incidence of pneumothorax
Data reporting the incidence of pneumothorax in VTV and control groups are described in eight trials (n=595).19 ,20 ,25 ,27 ,29 ,31 ,33 ,35 The meta-analysis demonstrated a significantly lower incidence of pneumothorax in preterm infants treated with VTV compared to those treated with PLV (RR 0.52 (95% CI 0.29 to 0.93), p=0.03). NNT was 20 (95% CI 11 to 100). There was no evidence of significant heterogeneity between studies (p=0.76, I2=0).
Failure of the mode of primary ventilation
Data reporting the failure of the primary mode of ventilation in VTV and PLV groups are described in four trials.19 ,27 ,31 ,33 The meta-analysis demonstrated that preterm infants treated with VTV had a significantly lower failure of primarily assigned mode of ventilation compared to those treated with PLV (RR 0.64 (95% CI 0.43 to 0.94, p=0.02), table 2). NNT was 11 (95% CI 6 to 50). There was no evidence of significant heterogeneity between studies (p=0.92, I2=0).
Meta-analysis results for secondary outcomes between volume-targeted ventilation and pressure-limited ventilation groups
Any episode of hypocarbia
Data reporting any episode of hypocarbia in VTV and PLV groups are described in two trials.18 ,24 The meta-analysis demonstrated that preterm infants treated with VTV had significantly fewer episodes of hypocarbia compared to those treated with PLV (RR 0.56 (95% CI 0.33 to 0.96), p=0.04). There was evidence of significant heterogeneity between studies (p=0.34, I2=0).
Supplemental oxygen administration
Data reporting supplemental oxygen administration in VTV and PLV groups are described in two trials.21 ,27 The meta-analysis revealed a mathematical MD of −1.68 (−2.47 to −0.88) days reduced supplemental oxygen administration using VTV compared to PLV (table 2). There was no evidence of significant heterogeneity between studies (p=0.60, I2=0).
Ventilatory parameters
Data reporting ventilatory parameters in VTV and PLV groups are described in seven trials.14 ,22 ,23 ,25 ,30 ,34 ,35 The meta-analysis revealed a mathematical MD of −0.54 (−1.05 to −0.02) cm H2O reduced MAP using VTV compared to PLV (table 2). There was evidence of low heterogeneity between studies (p=0.25, I2=24.7%). Meta-analysis did not show a difference in PIP, minute ventilation and inspired oxygen concentration between groups (table 2).
Other outcomes
Table 2 shows the comparison of other outcomes between preterm infants with VTV and PLV modes. These results were not significantly different between groups.
Discussion
Assessment of main results
VTV is an optional ventilation mode which has become the most widely and extensively studied in premature infants. During the last 10 years, ventilator software algorithms have been developed by combining PLV and volume targeting. Thus, sudden changes in lung compliance are detected and overcome by automatic adaptation of PIP. Target volume is achieved, ensuring more stability on discharged volumes. In recent systematic reviews, Wheeler et al15 ,16 reported that infants ventilated using VTV modes have improved outcomes compared to infants ventilated using PLV modes, including reduced death/BPD, duration of ventilation, pneumothoraces, hypocarbia, PVL and severe IVH. We have updated this evidence by performing a systematic review that includes an additional six studies.
The current review had a different search strategy and showed differences in outcomes compared with the previously published reviews.15 ,16 In our review, all outcomes pertaining to BPD or death were pooled together. The use of VTV was associated with a significant reduction in the incidence of BPD, but no reduction in the incidence of death. This result indicates that VTV may prevent the development of BPD, especially in very preterm infants who are most at risk for BPD development.38 One small trial showed that infants with severe RDS supported with pressure support ventilation+VG had lower proinflammatory cytokine levels in tracheal samples and lower MAP compared to those supported with pressure support ventilation.24 These data suggest that VTV is a potential and promising strategy for limiting lung inflammation and the development of lung injury.
In this review, we defined the length of mechanical ventilation as the primary outcome to measure the efficacy of VTV among preterm infants. Despite differences in ventilation mode and ventilators, birth weight, gestational ages and trial methodology, the effect sizes of the treatment with VTV reported by nine independent studies were similar. The pooled results from these nine trials demonstrated that VTV produced a 2-day reduction in the mean length of mechanical ventilation. Two other trials reported median values when describing the length of mechanical ventilation in VTV versus PLV groups20 ,27; therefore, these data were not included in our meta-analyses. This is a potential source of inaccuracy, and results should be interpreted appropriately.
The use of VTV was associated with a significant reduction in failure of the primary assigned mode of ventilation, any episode of hypocarbia, IVH, grade 3/4 IVH, pneumothorax, supplemental oxygen administration and PVL. These data are in accordance with the two previous systematic reviews of VTV versus PLV.15 ,16 We did not identify an increase in any adverse outcomes associated with the use of VTV compared with PLV.
In addition, we compared ventilator parameters between two groups. These results were not compared in the two previous systematic reviews.15 ,16 Lower PIP and MAP may limit barotraumas and reduce the incidence of CLD.39 An association between MAP levels and pneumothorax was observed.40 The use of VTV was associated with a significant reduction of −0.54 cm H2O MAP. This result provides insight when interpreting the lower incidence of BPD and pneumothorax in the VTV group. However, there were no differences in PIP, minute ventilation and inspired oxygen concentration between groups.
Heterogeneity of trial results
There was significant heterogeneity between various trials. Subgroup analysis was performed according to ventilation mode and ventilators, birth weight, gestational ages and trial methodology, but the presence of statistically significant heterogeneity remained in subgroups.
Limitations of our analysis
There are several limitations to our analysis. First, none of the studies included in this review attempted to blind the caregivers to group assignment. In the majority of studies, the allocated treatment method of each patient was known to those assessing the trial outcomes. Second, there is a lack of consistency in ventilation mode and ventilators, birth weight, gestational ages and methodology. Third, publication bias may exist. Finally, the studies included in this systematic review are diverse in parameter settings of ventilators and times at which studies were conducted, which may represent potential sources of bias.
Conclusion
Evidence from our systematic review indicates that preterm infants ventilated using VTV modes had a reduced incidence of BPD, length of mechanical ventilation, failure of primarily assigned ventilatory mode, IVH, grade 3/4 IVH, pneumothorax and PVL compared with preterm infants ventilated using PLV modes. Furthermore, multi-centre RCTs are required to evaluate improvements in neurodevelopmental outcomes, mortality and complications associated with mechanical ventilation.
References
Footnotes
-
Contributors WS Peng, HW Zhu, H Shi, and EM Liu were equally responsible for planning, conducting and reporting the work.
-
Competing interests None.
-
Provenance and peer review Not commissioned; externally peer reviewed.
Linked Articles
- Fantoms